| NOS Nitrous oxide (laughing gas) |
|
What Is Nitrous Oxide?
“I am sure the air in heaven must be this wonder working gas of delight”.
So wrote the poet Robert Southey of nitrous oxide, N2O, also known as nitrogen oxide, dinitrogen monoxide, hyponitrous acid anhydride, and factitious air. Its most well-known name, however, is “laughing gas” or “happy gas,” owing to its intoxicating effects when inhaled. At room temperature, it is a colourless, non-flammable gas with a pleasant, slightly sweet odour and taste.

Discovery and Early History
N2O was first discovered in 1793 by the English scientist and clergyman Joseph Priestley, who was also famous for being the first to isolate other important gases such as oxygen, carbon monoxide, carbon dioxide, ammonia, and sulphur dioxide. Priestley made N2O by heating ammonium nitrate in the presence of iron filings and then passing the resulting gas (NO) through water to remove toxic by-products.
The reaction he observed was:
2NO + H2O + Fe N2O + Fe(OH)2
After initial trials, Priestley thought that N2O could be used as a preserving agent, but this proved unsuccessful.
Following Priestley’s discovery, Humphry Davy of the Pneumatic Institute in Bristol, England, experimented with the physiological properties of the gas, particularly its effects upon respiration. He even administered the gas to visitors to the institute, and after watching the amusing effects on people who inhaled it, coined the term “laughing gas.”
Davy also noted the anaesthetic effects of the gas:
“As nitrous oxide in its extensive operation appears capable of destroying physical pain, it may probably be used with advantage during surgical operations in which no great effusion of blood takes place”.
Despite this observation, for the next 40 years or so the primary use of N2O was for recreational enjoyment and public shows. So-called nitrous oxide capers took place at travelling medicine shows and carnivals, where the public would pay a small price to inhale a minute’s worth of the gas. People would laugh and act silly until the effect wore off abruptly, at which point they would stand about in confusion. Many prominent figures from Clifton and Bristol came to inhale Davy’s purified nitrous oxide for recreational purposes.
Medical and Scientific Uses
Nitrous oxide found a more scientific use as an anaesthetic in clinical dentistry and medicine in the early 1840s. It remains a very safe and popular agent still utilised by dentists today. It is much less toxic than alternatives such as chloroform, with far less risk of explosion than ether. The main use for N2O is typically as a mild sedative and analgesic. It helps to allay the anxiety that many patients may have toward dental treatment, and it offers some degree of pain relief. However, when inhaled in pure form in large quantities, it will cause death by asphyxiation because at atmospheric temperatures and pressure, the oxygen in nitrous oxide is not available to the body.
Environmental Impact
At room temperature, N2O is quite unreactive with most substances, including alkali metals, halogens, and even ozone. Nitrous oxide reacts with ozone and is the main naturally occurring regulator of stratospheric ozone.
Nitrous oxide is also a major greenhouse gas. Considered over a 100-year period, it has 298 times more impact per unit weight than carbon dioxide. Although nitrous oxide makes up an extremely small proportion of the atmosphere – less than one-thousandth as abundant as carbon dioxide – it is 200 to 300 times more effective at trapping heat. It has one of the longest atmospheric lifetimes of the greenhouse gases, lasting for up to 150 years.
When heated sufficiently, N2O decomposes exothermically into N2 and O2. It is also used as an oxidiser in rocketry (this has the advantages over other oxidisers of being non-toxic and, due to its stability at room temperature, easy to store and relatively safe to carry on a flight; as a secondary benefit, it can be readily decomposed to form breathing air; its high density and low storage pressure enable it to be highly competitive with stored high-pressure gas systems) and in motor racing to increase the power output of engines. At elevated temperatures, nitrous oxide is a powerful oxidiser similar to oxygen.

How Nitrous Oxide Works in Engines
What does all this have to do with cars?
You have seen it in the movies. Hit a button and in an instant the engine produces 50, 100, 150, or more additional horsepower. Is it magic? Not really. But before understanding how a nitrous oxide system delivers that rush of additional power, it helps to understand a little about how the engine itself works.
Internal combustion engines are designed to convert one form of energy to another. The engine takes the energy stored in fuel and, through combustion, converts it into heat and pressure to produce power and torque at the flywheel. The key principle is simple: the more fuel that can be combusted, the more power that can be produced.
This brings us to combustion. What is needed to burn more fuel? Just two things. First, a way of adding additional fuel. Second, and more importantly, something that will supply an additional amount of oxygen to allow that fuel to burn.
In racing, nitrous oxide (often referred to as “nitrous” or “NOS,” from the brand name Nitrous Oxide Systems) allows the engine to burn more fuel with more oxygen, resulting in a more powerful combustion.
A key property of nitrous oxide is that at about 300 degrees Celsius it breaks down into nitrogen and oxygen. When it is introduced into the intake of an internal combustion engine, it is drawn into the combustion chamber and, on the compression stroke, when the charge air temperature reaches about 300 degrees Celsius, a very oxygen-rich mixture results. The injection of nitrous oxide into an engine therefore means that more oxygen is available during combustion. Because more oxygen is present, more fuel can – and must – also be injected, allowing the same engine to produce more power.
However, too much oxygen can become a problem. High levels of oxygen alone will cause detonation and engine damage. To keep things safe, the air/fuel ratio must be maintained, so additional fuel must be delivered whenever the nitrous system is active. Bigger fuel lines are a must, and a high-output fuel pump is also necessary.
The Cooling Effect
Nitrous oxide provides another performance benefit beyond the extra oxygen.
The gas is stored in a high-pressure tank at about 900 psi. At this pressure, nitrous oxide is in liquid form. When it is released into the intake manifold at atmospheric pressure, it changes to a gas, expands, and vaporises. This phase change provides a significant cooling effect on the intake air.
Reducing the intake air temperature increases air density, resulting in a denser charge that allows even more air/fuel mixture to enter the cylinder. The combination of the extra oxygen and the lower manifold temperature from the liquid-to-gas phase change creates a substantial power boost.
When extra fuel is added during nitrous oxide injection, the effect is similar to a supercharger or an increase in the engine’s compression ratio. Automotive nitrous systems work like the automotive equivalent of a jet’s afterburner and are used only for short-duration bursts of extra power. Nitrous oxide achieves this effect because it has a higher oxygen content by weight than atmospheric air: nitrous contains 36% oxygen by weight compared to the atmosphere’s 21 to 23%. Additionally, nitrous oxide is 50% more dense than air at the same pressure.
The Chemistry
The chemical reaction that occurs in the cylinder is as follows:
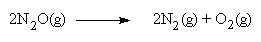
If this reaction occurs in the combustion chamber, extra oxygen is produced, providing an additional boost. There is a further benefit: the nitrogen itself buffers the increased cylinder pressure, helping to control the combustion process.
Typical NOS kit for stock car upgrade
Practical Limitations
Nitrous oxide is also an excellent value on a dollar-per-unit-power basis when installed and operated properly. The downside is that the fun ends quickly. The power boost lasts only as long as the bottle of nitrous. The average bottle holds 5 or 10 kilograms, and with a street V6 at 4,000 RPM that might provide 30 to 120 seconds of use.
Although it delivers impressive power gains for naturally aspirated engines, the rate of depletion is rapid. For this reason, the use of nitrous oxide is primarily confined to drag racing, street racing, and other short-duration racing applications. Maximum acceleration is the primary objective in these disciplines, while endurance is not. A car typically carries only a few minutes’ worth of nitrous oxide, and the driver uses it selectively by pushing a button.
Another drawback of a nitrous oxide system is that it is fairly bulky.
System Components
Regardless of the manufacturer, every nitrous oxide system accomplishes both tasks of supplying the gas and delivering an additional amount of fuel.
|
|
Dry system nozzle | |
Wet system nozzles |
In some way – either through the stock injectors or by adding additional fuel nozzles – the nitrous oxide control system puts more fuel into the engine.
The Basic System
A bottle, high-pressure lines, solenoids, jets, and nozzles are included in all nitrous oxide systems. The bottle usually holds 5 to 10 kilograms of nitrous oxide, and the boot (trunk) is the most popular mounting location.
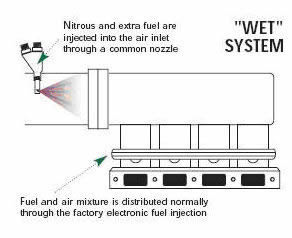
A high-pressure line carries the nitrous oxide to the solenoid valve. The solenoid is an electronically controlled valve. When a 12-volt signal is sent to the solenoid, it opens and nitrous oxide is directed to the nozzle and into the engine. At the same time, the system adds additional fuel by some means. The original and most simplified method of injecting the fuel is by using a solenoid and nozzle setup. This configuration is often referred to as a “wet” system and is nearly identical to the nitrous delivery arrangement, except that the fuel is injected at anywhere from 6 to 60 psi, while the nitrous oxide is injected at 700 to 1,200 psi.
|
|
Dry and Computer-Controlled Systems
“Dry” and “Computer Controlled” Systems
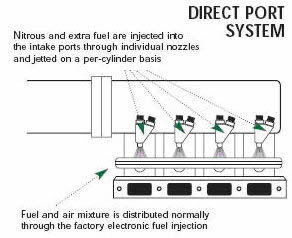
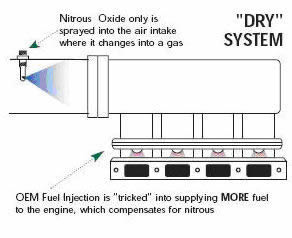
The term “dry” nitrous oxide system refers to systems that use the car’s original injectors to supply the additional fuel. The original dry systems designed by Nitrous Oxide Systems send a pressure signal to the stock fuel pressure regulator to increase fuel pressure. This increased pressure allows the injectors to supply more fuel for the added nitrous oxide.
Taking a slightly different approach, Venom’s VC-2000 system uses a computer control module to increase the amount of time that the injectors are open, thereby putting more fuel into the engine. Additionally, the Venom system uses the signal from the vehicle’s oxygen sensor to ensure that a safe air-fuel ratio is maintained. If the sensor detects an excess of nitrous without enough fuel, the Venom system shuts off the delivery of nitrous oxide, protecting the engine from damage.

High pressure braided hose lines
The Good, the Bad, and the Ugly
The Good, the Bad, the Ugly
The good news is that dollar for dollar, the horsepower increase from a properly installed nitrous oxide system is hard to beat. Most starter systems come ready to generate an additional 40 to 120 hp. Above this power level, a more sophisticated system is required and previous experience with nitrous oxide is highly recommended.
The bad news is that an improperly installed nitrous oxide system can cause severe engine damage. If nitrous oxide is injected into the engine without an adequate amount of fuel – or no fuel at all – temperatures in the combustion chamber will skyrocket, the engine may detonate, and parts will be broken and melted.
There are also some uncomfortable realities to face when using a nitrous oxide system:
First, bottles are only so big. The more frequently the system is used, the more often the bottle will need refilling.
Second, the less you learn about nitrous, the more likely you are to have a bad experience.
Third, and most important, it is not the use of nitrous oxide that causes engine damage. It is the misuse of nitrous oxide by the tuner and driver. Every engine has its limit. A stock engine doesn’t have the toughness of a racing engine. Chances are that there’s a tuner out there with your same engine and they have experience with using nitrous oxide on your engine. If they tell you that a 40 hp shot is all that you can do on a stock engine, believe them.
NOS solenoid valves
Safety Considerations
Better Safe Than Sorry
For a successful nitrous oxide experience, there are several considerations that can increase the chances of a positive outcome. Since a nitrous oxide system relies on the fuel system, it is always best to ensure that the fuel system is at peak efficiency. Factory fuel filters begin to degrade in performance as early as 10,000 kilometres.
When installing a new nitrous oxide system, it is recommended to replace the factory fuel filter and ensure that the injectors are clean.
The other area that must be addressed is the vehicle’s ignition system. A factory ignition system is designed to operate near factory horsepower levels. A nitrous oxide system can easily exceed the capabilities of a stock ignition system. Spark plugs should be new and ignition cables in good condition. The distributor cap and rotor should also be inspected on non-direct-ignition-system (non-DIS) cars. An ignition amplifier, high-performance ignition wires, and spark plugs one heat range cooler (for applications over 50 hp) are all advisable. In the absence of an ignition amplifier, it is a good idea to tighten the spark plug gap by 0.25 to 0.1 mm. This will make it easier for the ignition to generate a spark even in the high-horsepower range.
Health Risks and Legal Status
Because nitrous oxide is minimally metabolised, it retains its potency when exhaled into the room by the patient and can pose an intoxicating and prolonged-exposure hazard to clinic staff if the room is poorly ventilated. Where nitrous oxide is administered, a continuous-flow fresh-air ventilation system or nitrous-scavenging system is used to prevent waste gas buildup. Nitrous oxide can be habit-forming because of its short-lived effect.
In the United States, possession of nitrous oxide is legal under federal law and is not subject to DEA purview. It is, however, regulated by the Food and Drug Administration under the Food, Drug and Cosmetics Act; prosecution is possible under its “misbranding” clauses, prohibiting the sale or distribution of nitrous oxide for the purpose of human consumption.
In some countries, it is illegal to have nitrous oxide systems plumbed into an engine’s intake manifold. These laws are ostensibly used to prevent street racing and meet emission standards.
Nitrous oxide is legal to possess and inhale in the United Kingdom, although supplying it to others to inhale, especially minors, is more likely to result in prosecution under the Medicines Act.
In New Zealand, the Ministry of Health has warned that nitrous oxide is a prescription medicine, and its sale or possession without a prescription is an offence under the Medicines Act.
In Croatia, nitrous oxide is mentioned only in legislation concerning medical use, which would seemingly prohibit all non-medicinal applications.