Kevlar
Introduction
Kevlar is the registered trademark for a light, strong para-aramid synthetic fibre, related to other aramids such as Nomex and Technora.
Developed at DuPont in 1965 by Stephanie Kwolek and Roberto Berendt, it was first marketed in 1971. It is typically spun into ropes or fabric sheets that can be used as-is or as an ingredient in composite material components.
History of DuPont Innovation
Toward the end of the 1920s, the next important breakthrough for DuPont Corporation came through fundamental rather than applied research. The head of research noted at the time that they were including in the 1927 budget an item to cover pure science or fundamental research work, aimed at establishing or discovering new scientific facts.
In a short time, the research group developed an understanding of radical polymerisation and established the basic principles for condensation polymerisation and the structure of condensation polymers. This led to the invention and commercialisation of nylon in 1938, marking the beginning of the modern materials revolution. Prior to nylon, the group had invented neoprene synthetic rubber in 1933.
Many synthetic materials were subsequently invented by DuPont research, forming the basis for global businesses and products including well-known brands such as Teflon fluoropolymer resins, SilverStone non-stick finishes, Stainmaster flooring systems, Kevlar brand fibre, Nomex brand fibre and paper, Lycra spandex fibre, Sontara spun-laced fabric, Mylar polyester film, Tyvek spunbonded olefin, Cordura nylon fibre, and Corian solid surface material.

Properties and Applications
Kevlar has many applications, ranging from bicycle tyres and racing sails to body armour, owing to its high strength-to-weight ratio – famously described as five times stronger than steel on an equal weight basis.
A similar fibre called Twaron, with roughly the same chemical structure, was introduced by Akzo in 1978 and is now manufactured by Teijin.
Chemical Structure
Kevlar is synthesised from the monomers 1,4-phenylenediamine and terephthaloyl chloride in a condensation reaction that yields hydrochloric acid as a byproduct. The result is a liquid-crystalline behaviour, and mechanical drawing orients the polymer chains in the fibre’s direction.
Some other common synthetic polymers include nylon, Teflon, Lycra, and polyester. A polymer is a chain made of many similar molecular groups, known as monomers, that are bonded together.
The orientation of the polymer chains is very important to certain properties such as flexibility, rigidity, and strength. A group of polymer chains can be organised randomly in a pile, or oriented neatly side by side in a row.
Crystalline Structure
A Kevlar fibre is an array of molecules oriented parallel to each other, much like a package of uncooked spaghetti. This orderly, untangled arrangement is described as a crystalline structure. Crystallinity is obtained by a manufacturing process known as spinning, which involves extruding the molten polymer solution through small holes. The crystallinity of the Kevlar polymer strands contributes significantly to its unique strength and rigidity.
The individual polymer chains are held together by electrostatic forces between molecules known as hydrogen bonds.
When it comes to hydrogen bonds, Kevlar and water have something in common. In both compounds, the oxygen atoms have a high density of electrons around the nucleus, giving them a slight negative charge. Hydrogen atoms, on the other hand, have a much lower electron density, giving them a partial positive charge. Like the north and south poles of magnets, the positive hydrogen and negative oxygen of different molecules attract each other, forming hydrogen bonds.
Molecular Imaging
Scientists can use a special type of X-ray microscopy called XANES to reveal the orientation of molecules in materials. At the National Synchrotron Light Source in New York, scientists exposed the cut end of a Kevlar fibre to produce the following image:
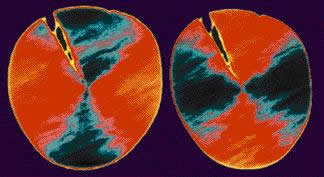
The pattern shows that the aromatic components of Kevlar have a radial (spoke-like) orientation.
This radial orientation is important because it allows the polymer chains to be well-ordered and symmetrical, like the atoms in a crystal. Because of this highly ordered structure, a fibre of Kevlar has only a few structural flaws or weak places. This lack of defects is the primary reason for the exceptional strength of Kevlar. Kevlar’s strength is five times that of steel, and its modulus is 75% that of carbon fibre.
Manufacturing
Kevlar (poly paraphenylene terephthalamide) production is expensive because of the difficulties arising from using corrosive concentrated sulfuric acid, which is needed to keep the water-insoluble polymer in solution during synthesis and spinning.
Kevlar is the most impact-resistant fibre on the market and also exhibits the highest tensile strength and greatest resistance to damage, vibration, and crack propagation.
However, Kevlar has very low compressive strength, so laminates carrying bending or compression loads should have carbon fibre incorporated to provide adequate stiffness. Kevlar is also hygroscopic, ultraviolet-sensitive, and cannot be sanded, making it unsuitable as an outer ply in a laminate. It is categorised by weight per square metre, available in a cloth weave, and sold by the running yard.

Types of Kevlar
Kevlar has different types of weaves. The weave that produces a fabric-like material for protective vests is called Kevlar 29. Kevlar 29 may also be used in brake lines or to replace asbestos. It is a major component of body armour.
Kevlar has two other primary types: Black Kevlar and Kevlar 49. Black Kevlar may be used to replace rubber items such as tyres. Kevlar 49 is extremely strong and can replace more traditional materials used for boat hulls or in items like bicycle frames.
Use in Formula 1
Kevlar is an excellent material for F1 bodywork and tyres. As a radial cord material, it works well for several reasons. In tyre construction, it is not necessarily a massive part of the entire tyre or cord. Bridgestone, Michelin, and Pirelli F1 tyres use a multi-layered, multi-composite cord approach, and Kevlar is used either as a pure layer or layered and interlayered as a composite within individual layers. Layered together in a tight construction, it holds up well to flex for the duration required, which is relatively short in an F1 tyre. It is also highly resistant to high temperatures and is commonly used in tyre technology as an anti-puncture layer, since it is the same material from which bulletproof vests are made. Additionally, Kevlar provides better fluctuation in the casing for improved variable damping effects from compound to compound.
Some parts of F1 bodywork must, by FIA rules, be covered with Kevlar. For example, wings must have a final layer made of Kevlar to hold the carbon fibre together in the event of a collision, preventing carbon fibre elements from breaking into small pieces. The internal layer of the survival cell (monocoque) must also be made of Kevlar to prevent front suspension members from penetrating the tub and injuring the driver’s legs. This rule was imposed after Michael Schumacher’s crash at Silverstone, when a left front suspension wishbone penetrated the monocoque and broke his leg.
In driver helmets, one or more layers are made from Kevlar to prevent penetration in the event of an incident.
